 | Info
Sheets |
| | | | | | | | | | | | | | | | | | | | | | | | |
 | Out-
side |
| | | | |
|
| | | | |
Result : Searchterm 'Liver MRI' found in 0 term [ ] and 3 definitions [ ] and 3 definitions [ ], (+ 19 Boolean[ ], (+ 19 Boolean[ ] results ] results
| previous 6 - 10 (of 22) nextResult Pages :  [1] [1]  [2 3 4 5] [2 3 4 5] |  | |  | Searchterm 'Liver MRI' was also found in the following services: | | | | |
|  |  |
| |
|

The company changed its name from Advanced Magnetics, Inc. to AMAG Pharmaceuticals, Inc. in July 2007.
AMAG Pharmaceuticals, Inc., a biopharmaceutical company, developed and manufactured organ-specific diagnostic contrast agents that provide clearer images during magnetic resonance imaging ( MRI) tests used to detect tumors and other abnormalities.
The company had two MRI related products on the market: Feridex I.V. (for the diagnosis of liver lesions) and GastroMARK (used for bowel and abdominal MR imaging). In November 2008, AMAG Pharmaceuticals, Inc. decided to discontinue the manufacturing of Feridex. The development of Combidex as a contrast agent for lymph disease has also been stopped.
The Company has now two commercial products: Feraheme® and GastroMARK®.
Feraheme® is the trade name of Ferumoxytol (formerly Code 7228) and is indicated for the treatment of iron deficiency anemia. Feraheme® is also being developed as a diagnostic agent for vascular-enhanced magnetic resonance imaging ( MRI) to assess peripheral arterial disease.
MRI Contrast Agents:
Contact Information
MAIL
AMAG Pharmaceuticals, Inc.
61 Mooney St.
Cambridge, MA 02138
USA
| |  | |
• View the NEWS results for 'AMAG Pharmaceuticals, Inc.' (7).
| | | | |  Further Reading: Further Reading: | | Basics:
|
|
News & More:
| |
| |
|  | |  |  |  |
| |
|

Founded in 1991, ImaRx Pharmaceutical Corp. designs, develops and markets pharmaceuticals for medical imaging ( MRI, ultrasound and computed tomography) for the radiological imaging industry.
ImaRx Pharmaceutical Corp., announced 1999 that it has been acquired by E.I DuPont de Nemours and Co., Inc.. The terms of the acquisition provide a royalty-free licensing arrangement with a newly-formed company, ImaRx LLC ("LLC"), to pursue and develop new products and technologies for drug and gene de livery independent from DuPont.
Yamanouchi Pharmaceutical Co. Ltd., ImaRx' licensee for Asian territories for this product, will continue to develop the product in Asia as DuPont's licensee. ImaRx LLC will have ownership of all other targeted and therapeutic products previously owned by ImaRx, including imaging products outside of diagnostic ultrasound imaging and two other imaging products, SonoRx® and LumenHance®, which are both FDA approved and licensed to BRACCO Diagnostics.
MRI Contrast Agents:
Contact Information
MAIL
ImaRx LLC
1635 East 18th Street
Tucson AZ 85719-6803
USA
| |  | | | |
|  | |  |  |  |
| |
|
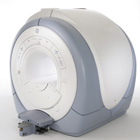
From GE Healthcare;
The Signa HDx MRI system is GE's leading edge whole body magnetic resonance scanner designed to support high resolution, high signal to noise ratio, and short scan times.
Signa HDx 3.0T offers new technologies like ultra-fast image reconstruction through the new XVRE recon engine, advancements in parallel imaging algorithms and the broadest range of premium applications. The HD applications, PROPELLER (high-quality brain imaging extremely resistant to motion artifacts), TRICKS (contrast-enhanced angiographic vascular lower leg imaging), VIBRANT (for breast MRI), LAVA (high resolution liver imaging with shorter breath holds and better organ coverage) and MR Echo (high-definition cardiac images in real time) offer unique capabilities.
Device Information and Specification CLINICAL APPLICATION Whole body
CONFIGURATION Compact short bore SE, IR, 2D/3D GRE, RF-spoiled GRE, 2DFGRE, 2DFSPGR, 3DFGRE, 3DFSPGR, 3DTOFGRE, 3DFSPGR, 2DFSE, 2DFSE-XL, 2DFSE-IR, T1-FLAIR, SSFSE, EPI, DW-EPI, BRAVO, Angiography: 2D/3D TOF, 2D/3D phase contrast vascular IMAGING MODES Single, multislice, volume study, fast scan, multi slab, cine, localizer H*W*D 240 x 2216,6 x 201,6 cm POWER REQUIREMENTS 480 or 380/415, 3 phase ||
COOLING SYSTEM TYPE Closed-loop water-cooled grad. | |  | | | |
|  |  | Searchterm 'Liver MRI' was also found in the following services: | | | | |
|  |  |
| |
|
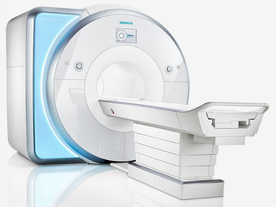
From Siemens Medical Systems;
Received FDA clearance in 2010.
MAGNETOM Skyra is a top-of-the-line, patient friendly wide bore 3 Tesla MRI system.
The system is equipped with the Tim 4G and Dot system (Total imaging matrix and Day optimizing throughput), to enhance both productivity and image quality with the complete range of advanced applications for clinical routine and research. Tim 4G features lighter, trimmer MRI coils that take up less space inside the magnet but de liver a high coil element density with increased signal to noise ratio and the possibility to use high iPAT factors.
Device Information and Specification
CLINICAL APPLICATION
Whole Body
Head, spine, torso/ body coil, neurovascular, cardiac, neck, shoulder, knee, wrist, foot//ankle and multi-purpose flex coils. Peripheral vascular, breast, shoulder.
CHANNELS (min. / max. configuration)
48, 64, 128
Chemical shift imaging, single voxel spectroscopy
MINIMUM TE
3D T1 spoiled GRE: 0.22 (256 matrix), Ultra-short TE
At isocenter: L-R 70 cm, A-P (with table) 55 cm
MAGNET WEIGHT (gantry included)
5768 kg
DIMENSION H*W*D (gantry included)
173 x 231 x 219 cm
COOLING SYSTEM
Water; single cryogen, 2 stage refrigeration
3 linear with 20 coils, 5 nonlinear 2nd-order
POWER REQUIREMENTS
380 / 400 / 420 / 440 / 460 / 480 V, 3-phase + ground; 110 kVA
| |  | | | |
|  | |  |  |  |
| |
|
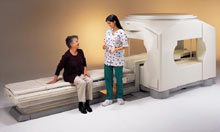
From Toshiba America Medical Systems Inc.;
the Ultra™ system was developed to help healthcare providers be more competitive by de livering greater patient comfort and a broad range of clinical capabilities, says Anita Bowler, product manager, MRI Business Unit, Toshiba America Medical Systems Inc With its unique, powerful gradient technology, the Ultra™ performs advanced clinical studies and consistently provides high-resolution images that are typically associated with high field MRI systems. At the same time, the Ultra™ offers a truly open feeling that makes patients more relaxed, especially those with claustrophobic tendencies.
Device Information and Specification CLINICAL APPLICATION Whole Body Quadrature, solenoid and multi-channel configurations SE, FE, IR, FastSE, FastIR, FastFLAIR, Fast STIR, FastFE, FASE, Hybrid EPI, Multi Shot EPI, Single shot EPI diffusion, True SSFP, SuperFASE; Angiography: 2D(gate/non-gate)/3D TOF, SORS-STC, Black Blood MRA POWER REQUIREMENTS 380/400/415/440/480 V COOLING SYSTEM TYPE Cryogenless | |  | | | |
|  | |  |  |
|  | |
|  | | |
|
| |
 | Look
Ups |
| |